ISO 13485 Lead Auditor
$1,890.00
Description
The PECB ISO 13485 Lead Auditor Certification course equips participants with the expertise to conduct Medical Devices Quality Management System (MDQMS) audits using recognized audit principles, procedures, and techniques. This training focuses on planning, executing, and managing both internal and external audits in compliance with ISO 19011 and ISO/IEC 17021-1. Designed for auditors, managers, consultants, and technical experts, the course aims to ensure conformance with MDQMS requirements, enhancing the delivery of safe and high-quality medical devices. By completing this course, participants gain the skills to lead audit teams, manage audit programs, and effectively communicate with stakeholders, preparing them for the certification exam and advancing their professional credentials.
Why Should You Attend the ISO 13485 Lead Auditor Course?
During this training course, you will acquire the knowledge and skills to plan and carry out internal and external audits in compliance with ISO 19011 and ISO/IEC 17021-1 certification process.
Based on practical exercises, you will be able to master audit techniques and become competent to manage an audit program, audit team, communication with customers, and conflict resolution.
Who Should Attend?
- Auditors seeking to perform and lead Medical Devices Quality Management System (MDQMS) certification audits
- Managers or consultants seeking to master a Medical Devices Quality Management System audit process
- Individuals responsible for maintaining conformance with Medical Devices Quality Management System requirements
- Technical experts seeking to prepare for a Medical Devices Quality Management System audit
- Expert advisors in Medical Devices Quality Management
Learning Objectives
- Understand the operations of a Medical Devices Quality Management System based on ISO 13485
- Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks
- Understand an auditor’s role to: plan, lead and follow-up on a management system audit in accordance with ISO 19011
- Learn how to lead an audit and audit team
- Learn how to interpret the requirements of ISO 13485 in the context of a MDQMS audit
- Acquire the competencies of an auditor to: plan an audit, lead an audit, draft reports, and follow-up on an audit in compliance with ISO 19011
Educational Approach
- This training is based on both theory and best practices used in MDQMS audits
- Lecture sessions are illustrated with examples based on case studies
- Practical exercises are based on a case study which includes role playing and discussions
- Practice tests are similar to the Certification Exam
Prerequisites
A fundamental understanding of ISO 13485 and comprehensive knowledge of audit principles.
ISO 13485 Lead Auditor Agenda
- Day 1: Introduction to Medical Devices Quality Management Systems (MDQMS) and ISO 13485
- Day 2: Audit principles, preparation and launching of an audit
- Day 3: On-site audit activities
- Day 4: Closing the audit
- Day 5: Certification Exam
ISO 13485 Lead Auditor Examination
- Domain 1: Fundamental principles and concepts of a Medical Devices Quality Management System (MDQMS)
- Domain 2: Medical Devices Quality Management System (MDQMS)
- Domain 3: Fundamental audit concepts and principles
- Domain 4: Preparation of an ISO 13485 audit
- Domain 5: Conducting an ISO 13485 audit
- Domain 6: Closing an ISO 13485 audit
- Domain 7: Managing an ISO 13485 audit program
Additional Information
- Training Days: 5
- CPD Certification (Credits): 31
- Exam Duration: 3 hour (open book)
- Free Retake Exam: Yes
- For more information, visit the PECB – ISO 13485 Lead Auditor Course page
Additional information
| Language | English, French (Français) |
|---|---|
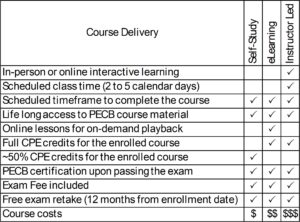
| Course Delivery | E-Learning, Self-Study |
Only logged in customers who have purchased this product may leave a review.





Reviews
There are no reviews yet.