ISO 13485 Foundation
$1,090.00
Description
The PECB ISO 13485 Foundation Certification course equips participants with the fundamental principles and requirements of a Medical Devices Quality Management System (MDQMS) as specified in ISO 13485. This training enables individuals to understand MDQMS policies, procedures, performance measurements, and continual improvement processes. The course is designed for those involved in medical devices quality management, individuals seeking knowledge about MDQMS processes, and those interested in pursuing a career in this field. By completing this course, participants gain a solid foundation in implementing and managing MDQMS, preparing them for the certification exam and enhancing their professional credentials.
Why Should You Attend the ISO 13485 Foundation Course?
ISO 13485 Foundation training enables you to learn the basic elements to implement and manage a Medical Devices Quality Management System (MDQMS) as specified in ISO 13485. During this training course, you will be able to understand the different modules of a MDQMS, including MDQMS policy, procedures, performance measurements, management commitment, internal audit, management review and continual improvement.
Who Should Attend?
- Individuals involved in Medical Devices Quality Management
- Individuals seeking to gain knowledge about the main processes of Medical Devices Quality Management Systems (MDQMS)
- Individuals interested to pursue a career in Medical Devices Quality Management
Learning Objectives
- Understand the elements and operations of a Medical Devices Quality Management System (MDQMS)
- Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks
- Understand the approaches, methods and techniques used for the implementation and management of a MDQMS
Educational Approach
- Lecture sessions are illustrated with practical questions and examples
- Practical exercises include examples and discussions
- Practice tests are similar to the Certificate Exam
Prerequisites
None
Agenda
- Day 1: Introduction to Medical Devices Quality Management System (MDQMS) concepts as required by ISO 13485
- Day 2: Medical Devices Quality Management System requirements and Certificate Exam
ISO 13485 Foundation Examination
- Domain 1: Fundamental principles and concepts of a Medical Devices Quality Management System (MDQMS)
- Domain 2: Medical Devices Quality Management System (MDQMS)
Additional Information
- Training Days: 2
- CPD Certification (Credits): 14
- Exam Duration: 1 hour (open book)
- Free Retake Exam: Yes
- For more information, visit the PECB – ISO 13485 Foundation Course page
Additional information
| Language | English, French (Français) |
|---|---|
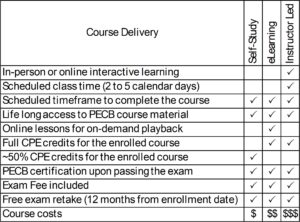
| Course Delivery | E-Learning, Self-Study |
Only logged in customers who have purchased this product may leave a review.





Reviews
There are no reviews yet.